 
These updated results will be featured in a late-breaking oral presentation at the American Society of Clinical Oncology (ASCO) 2023 Genitourinary Cancers Symposium from February 16-18, 2023. With a median follow-up of 36.1 months (31.6 months minimum), adjuvant Opdivo continued to show improved disease-free survival (DFS), non-urothelial tract recurrence-free survival (NUTRFS), distant metastasis-free survival (DMFS) and second progression-free survival (PFS2) compared to placebo across all-randomized patients and in patients whose tumor cells express PD-L1 ≥1%. Updated results from the Phase 3 CheckMate -274 trial will be presented in a late-breaking oral presentation at ASCO GU 2023īristol Myers Squibb (NYSE: BMY) today announced three-year follow-up results from the Phase 3 CheckMate -274 trial, demonstrating significant sustained clinical benefits with Opdivo (nivolumab) for the adjuvant treatment of patients with surgically resected, high-risk muscle-invasive urothelial carcinoma. placebo patients whose tumor cells express PD-L1 ≥1% remained disease-free more than six times as long vs. Randomized patients who received Opdivo after radical surgery remained disease-free more than twice as long vs. Three-year median follow-up data demonstrate significantly improved disease-free survival, non-urothelial tract recurrence-free survival, distant metastasis-free survival and second progression-free survival with adjuvant Opdivo compared to placebo These results support adjuvant NIVO as a new SOC for pts with MIUC with high risk for recurrence despite neoadjuvant chemo or those ineligible for and/or declining cisplatin-based chemo. AEs were manageable and consistent with previous reports. Conclusions: NIVO demonstrated a statistically significant and clinically meaningful improvement in DFS vs PBO for MIUC after radical surgery, both in ITT pts and pts with PD-L1 ≥ 1%. Grade 3–4 treatment-related adverse events (TRAEs) occurred in 17.9% and 7.2% of pts in the NIVO and PBO arms, respectively. DFS improvement with NIVO was generally consistent across subgroups. DFS and NUTRFS were improved with NIVO vs PBO in both populations (Table). 
The primary endpoint of DFS was met in ITT pts (median follow-up, 20.9 mo for NIVO 19.5 mo for PBO) and in pts with PD-L1 ≥ 1%. Results: In total, 353 pts were randomized to NIVO (PD-L1 ≥ 1%, n = 140) and 356 pts to PBO (PD-L1 ≥ 1%, n = 142). Non–urothelial tract recurrence-free survival (NUTRFS) in ITT pts and in pts with PD-L ≥ 1% is a secondary endpoint. DFS was stratified by nodal status, prior neoadjuvant cisplatin, and PD-L1 status. Primary endpoints: disease-free survival (DFS) in all randomized pts (ITT population) and in pts with tumor PD-L1 expression ≥ 1%. Pts had radical surgery within 120 days ± neoadjuvant cisplatin or were ineligible/declined cisplatin-based chemo, evidence of UC at high risk of recurrence per pathologic staging, were disease-free by imaging, and ECOG PS ≤ 1. Pts were randomized 1:1 to NIVO 240 mg Q2W or PBO for ≤ 1 year of adjuvant treatment. 
Methods: This is a phase 3, randomized, double-blind, multicenter trial of NIVO vs PBO in pts with high-risk MIUC (bladder, ureter, or renal pelvis) after radical surgery. This phase 3 trial of adjuvant nivolumab (NIVO) vs placebo (PBO) in pts with MIUC after radical surgery ± neoadjuvant cisplatin (CheckMate 274) aims to address an unmet need in these pts. There is no conclusive evidence supporting adjuvant chemo in pts who did not receive neoadjuvant chemo and in those with residual disease after neoadjuvant cisplatin. 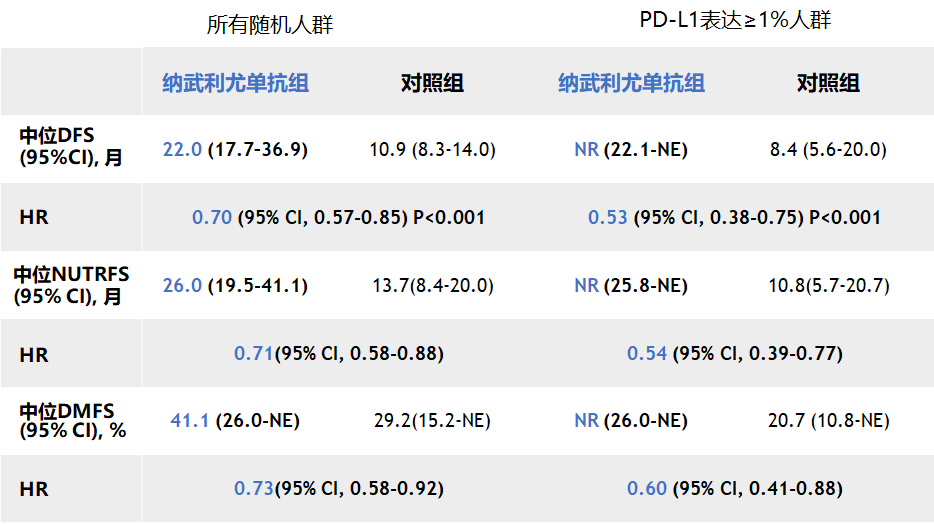
Background: The standard of care (SOC) for patients (pts) with MIUC is radical surgery ± cisplatin-based neoadjuvant chemotherapy (chemo), but many pts are cisplatin-ineligible. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
